
Because Sr-90 acts like calcium, milk monitoring will be important after a large release. Once in the body, Sr-90 acts like calcium and is readily incorporated into bones and teeth, where it can cause cancers of the bone, bone marrow, and soft tissues around the bone. Strontium-90 can be inhaled, but ingestion in food and water is the greatest health concern. No significant amount of Sr-90 reached the United States from either incident. Learn more about EPA’s historical responses to radiological emergencies like Fukushima and Chernobyl on the Planning and Past Responses page. Strontium-90 was also released during the 2011 Japanese nuclear incident at the Fukushima Daiichi Nuclear Power Plant. Now, everything weve found points to elements that formed only in the presence of lots of neutrons. The accident at the Chernobyl nuclear power plant introduced a large amount of Sr-90 into the environment. Strontium-90 is also found in waste from nuclear reactors. from the atmospheric testing of nuclear weapons. Strontium-90 was widely dispersed in the 1950s and 1960s in fallout falloutRadioactive material in the air from a nuclear explosion that will cool into dust-like particles and fall to the ground. Atomic nuclei would have snapped up more neutrons to become elements heavier than strontium. Strontium SourcesĮveryone is exposed to small amounts of Sr-90, since it is widely dispersed in the environment and the food chain. neutrons, the r-process would not have yielded strontium, which has relatively few neutrons in its nucleus. Animals may ingest Sr-90 when eating plants. Strontium S Element 38 Mass Number: 88 Atomic weight: 87.62 g/mol Atomic number (Z): 38 Electrons: 38 Protons: 38 Neutrons: 50 Period: 5 Group: 2 Block: s. Plants or crops growing in or near contaminated soil may take up small amounts of Sr-90 from the soil. 
is the only way of decreasing the amount of Sr-90 in the environment. It has been slowly decaying since then, current levels from these tests are very low. Radioactive decay Radioactive decayThe process in which an unstable (radioactive) nucleus emits radiation and changes to a more stable isotope or element. This fact has key implications for the building up of the periodic table of elements.Strontium-90 is widely dispersed in the environment and the food chain from atmospheric testing of nuclear weapons in the 1950s and 1960s. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. Strontium is most similar chemically to the heavier alkali earth elements, Calcium, and Barium. In the periodic table, the elements are listed in order of increasing atomic number Z. It has an atomic number of 38, an atomic mass of 88, one oxidation state (+2) and four naturally occurring isotopes ( 84 Sr, 86 Sr, 87 Sr, 88 Sr), of which 88 Sr is the most abundant at 82.6 of the total mass. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. This process gives a way to age rocks, by measuring the isotopes of rubidium and strontium with mass spectrometry, then calculating the ratios of the radioactive forms to. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The radioactive isotope decays to form strontium-87. Yttrium is a chemical element with atomic number 39 which means there are 39 protons in its nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Atoms of the same element with different numbers of neutrons. 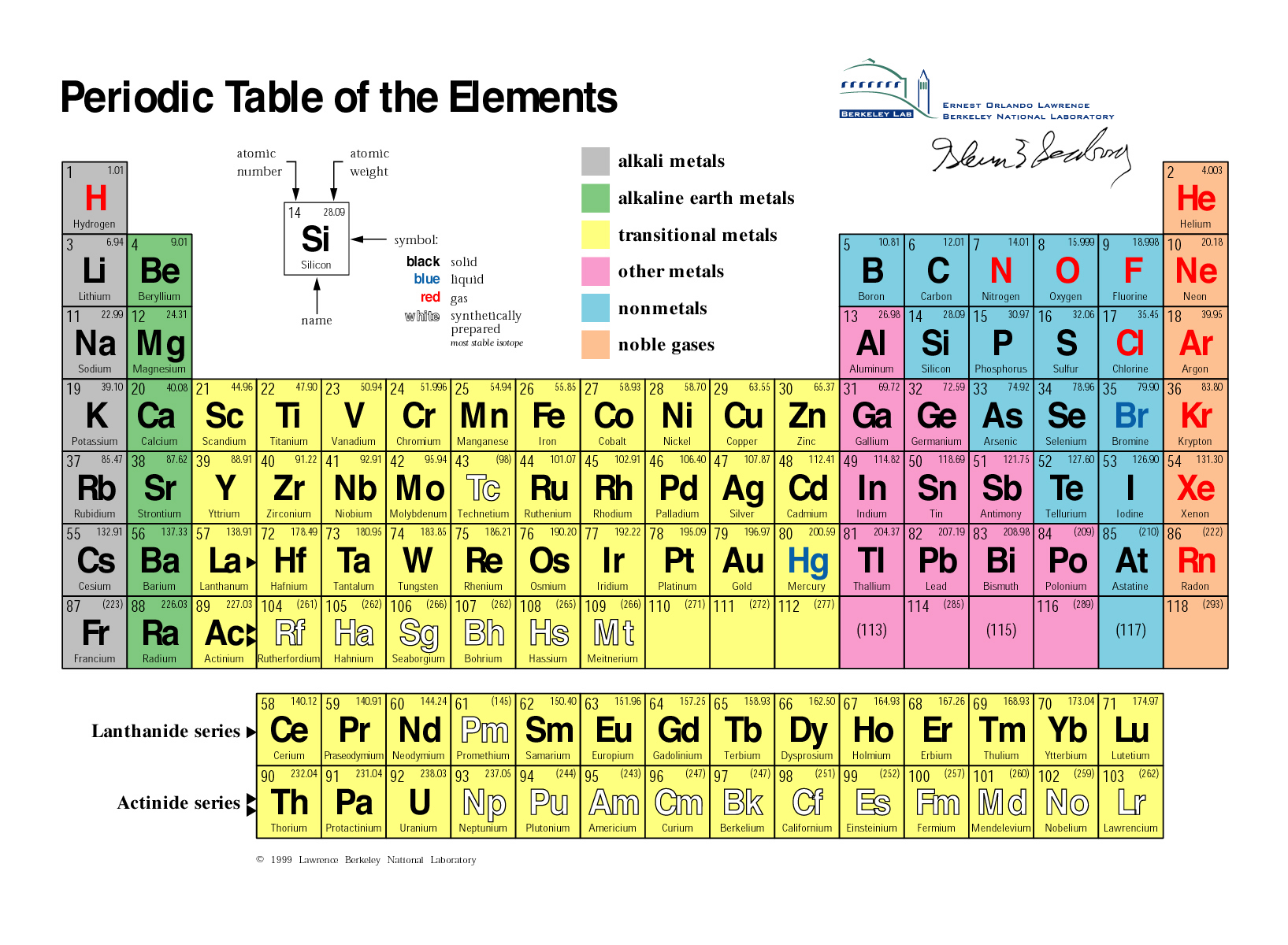
Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
